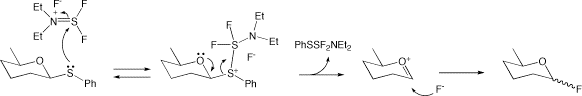Equipments & Technologies
Glycosyl fluoride synthesis with no sweat
Glycosyl fluoride is recognized highly in the synthesis of complex oligosaccharides. Unusual stability of the species compared to other glycosyl halides makes it handy donor, which is readily activated by Hafnocene ditriflate leading to generally good to excellent results.
The synthesis is usually based on the activation of thio-glycosides such as phenylthio-glycoside by bromonium ion species then reacted with fuloride anion generated from diethylaminosulfur trifluoride (DAST). The reaction is usually okay as it is, but always accompanies glycosyl bromide as a by-product. The amount of the bromide varies compound by compound.
Here is a trick to overcome this problem. “The reaction does not need bromonium ion.” Yes, DAST exists in equilibrium with sulfiminium fluoride and thus acts as both electrophile and nucleophile. Although reactivity is somewhat weaker than N-bromosuccinimide (NBS), reaction of phenylthio-glycoside and DAST proceeds just fine by slightly elevating reaction temperature. Moreover, the reaction is VERY clean giving high yield.
Typical protocol.
3,4,6-Tri-O-benzyl-2-O-chloroacetyl-α,β-D-galactopyranosyl fluoride.
To a solution of phenyl 3,4,6-tri-O-benzyl-2-O-chloroacetyl-1-thio-β-D-galactopyranoside (213.5 mg, 345 μmol) in CH2Cl2 (3.5 mL) was added DAST (85 μL, 69 μmol) at 0˚C and stirred at 40˚C for 3.5 h. The reaction mixture was diluted with EtOAc and extracted with saturated aq. NaHCO3 and brine. The organic layer was dried over Na2SO4, filtered, and concentrated. The residue was chromatographed on silica gel (hexane : EtOAc = 5:1) to give the fluoride (181.5 mg, 99.5 %, α / β = 1.00/0.32) as a pale yellow syrup.
References
-
Suzuki, K. Ito, Y. Kanie, O
-
Carbohydr Res 2012, 359, 81-91.
-
-
2.A method of orthogonal oligosaccharide synthesis leading to combinatorial library based on stationary solid-phase reaction.
-
Ako, T. Daikoku, S. Ohtsuka, I. Kato, R. Kanie, O.
-
Chem. Asian J., 2006, 1, 798-813.
-
Ohtsuka, I. Ako, T. Kato, R. Daikoku, S. Koroghi, S. Kanemitsu, T. Kanie, O.
-
Carbohydr. Res., 2006, 341, 1476-1487.
-
Kanie, O. Ohtsuka, I. Ako, T. Daikoku, S. Kanie, Y. Kato, R.
-
Angew. Chem. Int. Ed., 2006, 45, 3851-3854.
2010/10/29